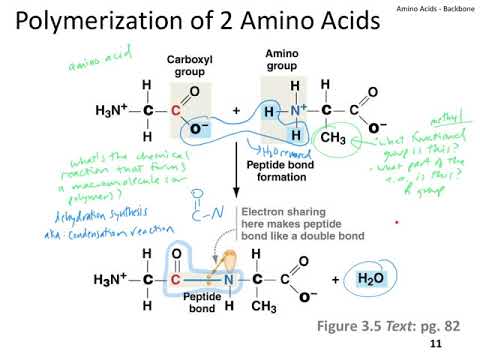
Although such a computational cost may not be much of a burden for on-site, non-interactive sequence design tasks targeting a few fixed backbones, it is expensive enough to hinder remote or online interactive applications, such as using ABACUS to design or analyze sequences through a web server. core to construct the single residue and the residue pairwise energy tables for a single target backbone, the actual computational cost being dependent on the size and shape of the target. It takes hours of a single central processing unit (CPU). This needed to be carried out for every backbone position and every position pair, which was time consuming, resulting in suboptimal computational efficiency. In the original ABACUS, the templates were identified by searching over the entire training set, which contains millions or more entries of backbone positions (or position pairs). In these senses, the method is a de novo one in terms of sequence design under given backbones ( Liu and Chen, 2016). When ABACUS is applied to design amino acid sequences, the only absolutely required input is a target backbone structure (which does not have to be a naturally existing one), while the computation process does not refer to any pre-existing target-specific sequence information, including sequence information from homologous proteins or structurally similar proteins. Because of this, the applicability of the statistically-derived ABACUS model is not restricted to proteins of particular overall sequence or structure families. Each energy term in ABACUS is expected to represent information contained in such basic units extracted from proteins of diverse overall sequences and structures. The above templates can be viewed as small, basic units of protein structures and sequences, each unit involving only one or two backbone positions. These training backbone position or position pairs will be called templates. The actual derivation of the energies associated with a particular target backbone position or position pair involves first finding training backbone positions (for the single residue energy) or position pairs (for the residue pairwise energy) that are close to the target in the space spanned by the chosen structural features, and then analyzing the amino acid compositions at these positions or position pairs. Likewise, the pairwise energy between two coupled backbone positions simultaneously depends on the relative geometries between the two positions as well as on the local structural and environmental features of the individual positions composing the pair. the Ramachandran angles and the secondary structure type) as well as the solvent accessibility of that position. More specifically, the single residue energy associated with a backbone position simultaneously and non-additively depends on the local conformation (i.e. One important characteristic of the statistical energy terms in ABACUS is that the dependence on different types of structural features is considered jointly in single terms. 
The ABACUS energy function is composed of single residue terms, residue pairwise terms and atomic packing terms ( Xiong et al., 2014 Zhou et al., 2016). 
For example, in comparison with the well-known RosettaDesign program ( Leaver-Fay et al., 2011), ABACUS usually provides alternative design results (sequence identity of about 30%) for the same target ( Xiong et al., 2014). Because of its distinct energy function, ABACUS can find solutions located in regions in the sequence space that are different from those explored by other protein design programs. It comprises a structure-dependent sequence energy function with mainly statistically-derived terms ( Sun and Kim, 2017 Topham et al., 2016 Wang et al., 2018 Xiong et al., 2014). The ABACUS (a backbone-based amino acid usage survey) method is one such tool, sequences designed using ABACUS having been experimentally verified to fold into expected structures of different fold types ( Xiong et al., 2014, 2017 Zhou et al., 2016). Primary tools for computational protein design include automated sequence design programs that can identify amino acid sequences compatible with given polypeptide backbone structures ( Alford et al., 2017 Dahiyat and Mayo, 1997 Gainza et al., 2013 Huang et al., 2016 Liu and Chen, 2016 O'Connell et al., 2018 Ollikainen et al., 2013 Simonson et al., 2013 Wang et al., 2018).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
